Global Intraosseous Infusion Devices Market to Reach USD 11.2 Billion by 2036, Driven by Emergency Care Demand
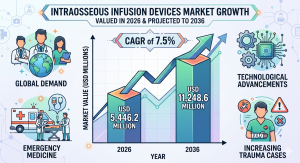
DE, UNITED STATES, April 1, 2026 /EINPresswire.com/ -- The global intraosseous infusion devices market is entering a steady growth phase, projected to expand from USD 5,446.2 million in 2026 to USD 11,248.6 million by 2036, registering a CAGR of 7.5%, according to Future Market Insights (FMI). The market growth is being fueled by the rising demand for rapid vascular access in emergency and critical care settings, increasing trauma incidence, and adoption of advanced devices with safety and procedural efficiency features.
Intraosseous infusion devices—designed for quick and reliable access to the vascular system—are becoming essential in prehospital care, emergency departments, trauma centers, and pediatric care, enabling life-saving medication and fluid delivery when conventional intravenous access is challenging.
Intraosseous Infusion Devices Market Quick Stats Snapshot:
• 2026 Market Value: USD 5,446.2 Million
• 2036 Forecast Value: USD 11,248.6 Million
• CAGR (2026–2036): 7.5%
• Leading Product Category: EZ-IO (21.5% share)
• Top Route of Administration: Sternum (30.9% share)
• Key End User: Emergency Medical Services (30.1% share)
• Key Growth Driver: Rising emergency care demand and trauma incidence
• Innovation Focus: Powered insertion devices, ergonomic designs, standardized training protocols
Market Momentum: Rapid Vascular Access Needs Driving Adoption
The intraosseous infusion devices market is expanding rapidly as emergency medical services and hospitals respond to the urgent need for rapid vascular access in trauma, cardiac arrest, shock, and pediatric emergencies. Adoption is driven by:
• Increased emergency care volumes and prehospital interventions
• Integration of intraosseous devices in advanced life support protocols
• Training and simulation-based education improving procedural confidence
• Regulatory approvals and standardized device guidelines
Product and Route Insights: Precision and Speed in Emergency Care
Product Leadership: EZ-IO leads with 21.5% value share due to powered insertion and predictable placement. Other products include B.I.G, FAST1, FASTR, IO needles, and specialized alternatives.
Route of Administration: Sternum accounts for 30.9% share, favored for rapid access to central circulation; other routes include distal/proximal tibia, distal femur, calcaneus, and humeral head.
End-User Dominance: EMS units command 30.1% share, with hospitals, ambulatory surgical centers, and cardiac care centers contributing significant adoption in controlled clinical environments.
Regional Insights: Growth Across Developed and Emerging Markets
India: Rapid growth (CAGR 8.7%) driven by rising trauma cases, ambulance network expansion, and emergency preparedness.
South Korea: CAGR 8.2%, led by high protocol compliance and advanced emergency systems.
Germany: CAGR 8.0%, supported by mature emergency frameworks and reimbursement structures.
France: CAGR 7.8%, fueled by structured emergency response systems and hospital-based critical care.
China: CAGR 7.6%, enabled by hospital network expansion and domestic manufacturing of cost-efficient devices.
North America & Europe: High adoption supported by advanced EMS, hospital infrastructure, and standardized emergency protocols.
Competitive Landscape: Innovation and Reliability at Core
The intraosseous infusion devices market is moderately consolidated, with Tier 1 players leading adoption through global distribution, clinical training programs, and robust device portfolios. Key players include:
• Teleflex
• Smiths Medical
• Becton Dickinson (BD)
• ICU Medical
• Zhejiang Shanshan Broadmed
Key Growth Drivers and Challenges
Drivers:
• Rising trauma incidence and emergency care demand
• Integration in prehospital and hospital protocols
• Advancements in powered insertion and ergonomic device designs
• Standardized training improving clinician confidence
Challenges:
• High cost of devices and single-use components
• Regulatory clearance and certification requirements
• Risk of improper placement or procedural complications
Strategic Takeaways for Decision Makers
• Expand adoption in high-trauma regions and emerging markets
• Invest in ergonomic and powered device innovation
• Strengthen EMS and hospital training programs
• Focus on integration with advanced life support protocols
Request Sample Report | Customize Report | Purchase Full Report:https://www.futuremarketinsights.com/reports/sample/rep-gb-1569
Why This Market Matters Now
The convergence of rising emergency care needs, trauma prevalence, and advanced device innovation is transforming critical care delivery. Intraosseous infusion devices enable faster, reliable vascular access, improving patient survival rates and procedural efficiency. For stakeholders, this market represents a high-value, technology-driven growth opportunity over the next decade.
Frequently Asked Questions (FAQ)
How big is the market in 2026? USD 5,446.2 million
What is the 2036 forecast? USD 11,248.6 million
What is the growth rate? 7.5% CAGR (2026–2036)
Which product leads? EZ-IO (21.5% share)
Which route dominates? Sternum (30.9% share)
Which end user leads? EMS (30.1% share)
Explore More Related Studies Published by FMI Research:
Non-Alcoholic Steatohepatitis Clinical Trials Market-https://www.futuremarketinsights.com/reports/non-alcoholic-steatohepatitis-clinical-trials-market
Aesthetic Medical Device Market-https://www.futuremarketinsights.com/reports/aesthetic-medical-devices-market
Malaria Treatment Market-https://www.futuremarketinsights.com/reports/malaria-treatment-market
IgA Nephropathy Market-https://www.futuremarketinsights.com/reports/iga-nephropathy-market
Chronic Wound Care Market-https://www.futuremarketinsights.com/reports/chronic-wound-care-market
Podiatry Services Market-https://www.futuremarketinsights.com/reports/podiatry-services-market
Aquaculture Vaccines Market-https://www.futuremarketinsights.com/reports/aquaculture-vaccines-market
About Future Market Insights (FMI)
Future Market Insights, Inc. (FMI) is an ESOMAR-certified, ISO 9001:2015 market research and consulting organization, trusted by Fortune 500 clients and global enterprises. With operations in the U.S., UK, India, and Dubai, FMI provides data-backed insights and strategic intelligence across 30+ industries and 1200 markets worldwide.
Why FMI: Decisions that Change Outcomes- https://www.futuremarketinsights.com/why-fmi
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware – 19713, USA
T: +1-347-918-3531
Website: https://www.futuremarketinsights.com
LinkedIn| Twitter| Blogs | YouTube
Have a specific Requirements and Need Assistant on Report Pricing or Limited Budget please contact us - sales@futuremarketinsights.com
Sudip Saha
Future Market Insights Inc.
+1 347-918-3531
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.