NIH Global Study: Histologics Gentle Kylon® Biopsy Brush Shows Diagnostic Advantages in Cervical Cancer Workups.
A sizeable minority of cases are endocervical, and the Soft-Brush biopsy sampler for the endocervix apparently increased the yield of precancer. This merits further examination.”
ANAHEIM, CA, UNITED STATES, April 30, 2026 /EINPresswire.com/ -- Histologics LLC, based in Anaheim California attended the Annual Meeting of the American Society of Colposcopy and Cervical Pathology in Orlando, FL from April 23-26, 2026. Mark Schiffman MD, MPH, Epidemiologist and Senior Investigator at the National Cancer Institute / National Institutes of Health led a session addressing efficient screening, triage and treatment of diagnosed high grade cervical cancer precursors performed during the “Moonshot” funded NIH PAVE (Human Papillomavirus (HPV)-Automated Visual Evaluation) study) performed in countries with significant cervical cancer death rates: Brazil, Cambodia, Dominican Republic, El Salvador, Eswatini, Honduras, Malawi, Nigeria, and Tanzania.— Dr. Mark Schiffman MD, MPH
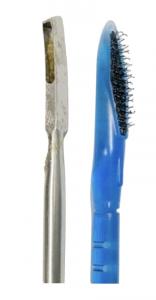
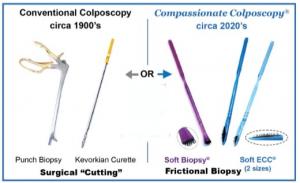
The study deployed several novel technologies in ascertaining risk by having patients self-collect vaginal swab tests for high-risk HPV, screen-positive cases undergoing diagnosis with a colposcope capable of artificial intelligence (IRIS, Liger Medical, Lehi, Utah), and removing tissue from the cervix for histologic “biopsy” testing deploying a proprietary medical sterile hooked biopsy nylon brush (Kylon®). Kylon® coated biopsy devices remove and trap abundant samples to pathology labs. (SoftBiopsy® for outer cervical lesions, Soft-ECC® for lesions from inside endocervical canal, where a visual test such as colposcopy cannot detect them).
Dr. Schiffman posed this aim in his presentation: “It is critical we design better tests to define precancer.” A new finding was that a larger amount of disease is hidden in the endocervical canal than previously documented with research that utilized conventional stainless steel curette devices for shaving tissue from the canal. In the PAVE clinical sites, some decided to utilize steel punch biopsy curettes while many sites chose Kylon® brush biopsy tools and Dr. Schiffman concluded “A sizeable minority of cases are endocervical, and the Soft-Brush biopsy sampler for the endocervix apparently increased the yield of precancer. This merits further examination”. The PAVE trial is planned to be extended to new countries and will utilize the Kylon® brushes as well.
Neal Lonky MD, MPH, public health and cervical cancer prevention expert, Founder and CEO of Histologics LLC, Clinical Professor of Obstetrics and Gynecology at the University of California Irvine, Former Elected Member of the Board of Directors of the Southern California Permanente Medical Group, and multi-patented inventor of the Kylon® device technology stated “My focus was to invent a device and method where removal of tissue for biopsy is more compassionate, easy to use, safe, and affordable. Histologics LLC and other independent non-profit funding sources have conducted research to support the high-quality outcomes and allay fears of the procedure with what we trademarked as Compassionate Colposcopy®. With approximately 2 million of our safe and sterile minimally invasive brush-biopsy devices used in the USA in nearly every clinical setting by trained physicians and nurse practitioners and processed in nearly every pathology laboratory setting. We want this to be effective, efficient, patient friendly, and cost effective and we are seeing this adoption growing. I am also thrilled that the American Society of Colposcopy and Cervical Pathology has incorporated our devices and methodology in the training courses and workshops on how to perform reliable and reproducible diagnostic workups after abnormal screening tests during colposcopy. This is transforming the standard of care.”
About Histologics
Histologics is medical device manufacturer with a mission to compassionately save lives, and is dedicated to advancing gentle, effective tissue management solutions for women’s health, advanced wound care, and veterinary medicine. Its proprietary, patented, and award-winning Kylon® technology platform is a hooked brush array medical fabric that can safely and compassionately be used to precisely and gently remove tissue from the body for diagnostics and therapeutic benefits. Histologics provides clinicians with innovative cost-effective tools that enhance procedural efficiency, accuracy, and patient comfort. Histologics products have been adopted by leading academic, government, HMO, hospitals, private practice settings, and home settings nationwide.
A compendium of research and clinical evidence over the last 15 years can be found at: https://www.histologics.com/resources/research-summary.html
For more information, visit https://histologics.com or contact Histologics Women’s Health Division at 888-738-9757.
Lily Ramos
Histologics LLC
+1 888-235-2275
email us here
Visit us on social media:
LinkedIn
Facebook
TikTok
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.